A Trusted Clinical Site Network Partner of the RAMAAYANAM Group
RAMAAYANAM Life Sciences


Who we are
RAMAAYANAM LIFE SCIENCES is a specialized Clinical Research Partner and Site Network Management within the RAMAAYANAM Group of Companies, delivering efficient, compliant, and high-quality clinical trial execution across India and global markets.
With strong on-ground operational capabilities and deep regulatory expertise, RAMAAYANAM LIFE SCIENCES supports end-to-end clinical development programs—from feasibility assessment to study close-out—ensuring timely, reliable, and quality-driven outcomes for sponsors and CRO partners.
Key Highlights
GCP-trained investigators and site teams.
12000+ global clinical sites database.
40000+ Participants database.
Regulatory approved BA/BE Centres.
Smart site activation.
Proven experience in local and global clinical trials.
Robust patient recruitment and retention capabilities.
Regulatory-compliant, quality-focused operations.
Flexible engagement models for sponsors and CROs.
Our Core Services
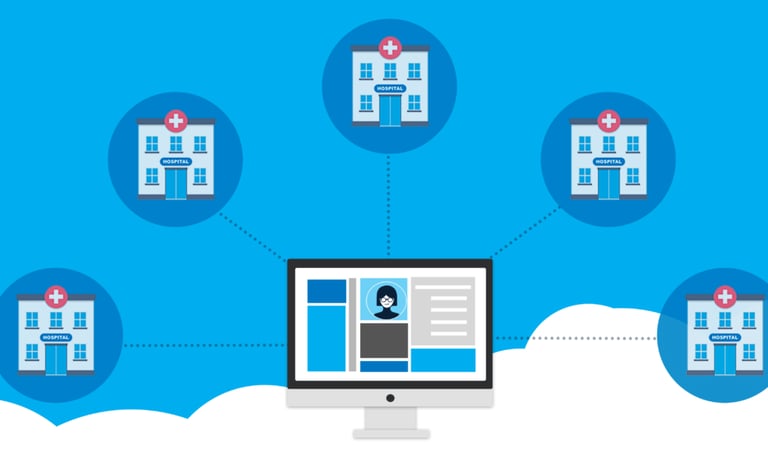








Site Management
Site identification, feasibility assessment, and selection.
Site activation and start-up support.
Investigator and site staff coordination.
Study conduct oversight and site monitoring support.
Patient recruitment and retention strategies.
Site close-out and essential document archival support.
Clinical Trial Execution Support
Phase I–IV clinical trials.
BA/BE and post-marketing studies.
Observational and real-world evidence studies.
Investigator-Initiated Trials (IITs).
Regulatory & Ethics Support
Ethics Committee (EC) submissions and approvals
Regulatory documentation coordination
Compliance with ICH-GCP, CDSCO, and global regulatory guidelines
Data & Quality Oversight
Source document preparation and review support
Query resolution coordination
Quality checks and audit readiness
CAPA management assistance
Project & Vendor Coordination
Study timelines and milestone tracking
CRO, laboratory, and vendor coordination
Budget tracking and site payment management
Vendor Management
Our Expertise
Advancing Research Across Multiple Specialties
We supports clinical research across a broad range of therapeutic areas, leveraging an experienced investigator network and patient-centric site management approach.
Cardiovascular diseases, hypertension, and long-term outcome studies.
Oncology
Cardiology
Neurology
Gastroenterology
Infectious Diseases
Endocrinology & Metabolic Disorders
Diabetes, thyroid disorders, and metabolic syndromes.
Vaccine and antiviral clinical trials.
Liver diseases, IBD, and digestive disorders.
Dermatology
Psychiatry
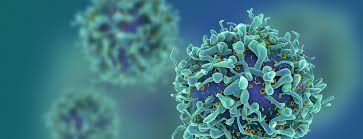
Immunology


Patient Engagement & Recruitment Support
Patient identification through site networks and referral pathways
Recruitment feasibility analysis and enrollment forecasting
Patient engagement, education, and retention programs.
Support for decentralized and hybrid clinical trial models.
Patient follow-up coordination and visit adherence monitoring


Major Depressive Disorder, schizophrenia, Bipolar Disorder, and anxiety disorders
Atopic dermatitis (eczema), psoriasis, hidradenitis suppurativa, acne vulgaris, and alopecia areata
Rheumatoid arthritis, lupus, IBD, psoriasis, and multiple sclerosis


Food and Beverages


Nutraceutical Supplements
Experienced in managing studies for dietary supplements, herbal products, and wellness formulations
Our network enables assessment of safety, tolerability, and health benefits in real-world populations
Solid tumors, hematologic malignancies, and immuno-oncology studies.
Alzheimer’s, Parkinson’s, epilepsy, stroke, depression, and schizophrenia
