How In Silico Trials and Digital Twins are Redefining the Future of Drug Development
Drug development is undergoing a quiet transformation. Instead of relying solely on traditional experimentation, researchers can now evaluate therapies within highly sophisticated digital models of human biology. How In Silico Trials and Digital Twins are Redefining the Future of Drug Development
Gaurav Singh
4/30/20264 min read
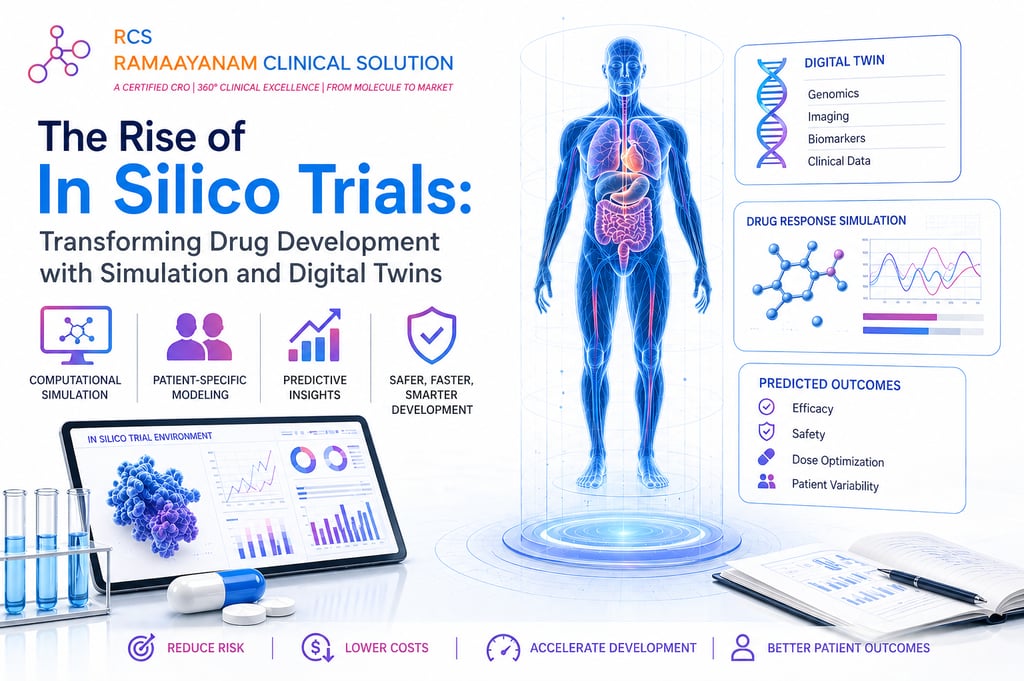
What Exactly Is an In Silico Trial?
The term “in silico” a playful nod to the silicon chips powering modern computers refers to experiments conducted entirely through computational models. In pharmaceutical research, this means building detailed mathematical representations of human organs, cells, and even individual proteins, then exposing these virtual systems to candidate drug molecules to observe how they react. Unlike traditional trials, no biological specimen is harmed, no petri dish is needed, and results can arrive in hours rather than months [2], [3].
Enter the Digital Twin
At the heart of this revolution is the concept of the digital twin a living, breathing computational replica of a specific patient or organ. Fed with real patient data such as genomics, imaging scans, and biomarker profiles, a digital twin can mirror individual physiology with remarkable fidelity. Researchers can then ask: “How will this 62-year-old diabetic patient’s heart respond to Drug X?” and get a statistically grounded answer before any real-world exposure occurs [4], [5].
Why This Changes Everything
Drug development is brutally expensive and slow. On average, bringing a new medicine to market takes over a decade and costs upward of $2.5 billion, with failure rates exceeding 90% in clinical phases [1], [2]. In silico trials can front-load the screening process, discarding toxic or ineffective compounds early long before costly human trials begin. Beyond economics, they promise something more profound: the ability to simulate rare populations, elderly patients, or those with comorbidities who are routinely underrepresented in conventional studies
With growing industry adoption and ongoing validation efforts, digital twins are rapidly becoming a powerful tool for more precise and personalized drug development.
How RCS Is Bridging the Gap Between Simulation and Clinical Reality
At RCS, we are actively integrating simulation-driven approaches into the way drug development is designed and delivered. As a full-service Contract Research Organisation, we bring together clinical expertise, advanced analytics, and technology enabled modeling to ensure that in silico insights directly inform real-world studies.
We apply predictive modeling and simulation techniques to evaluate dose response, safety signals, and endpoint behaviour early in the development process. By working with structured clinical datasets, real-world evidence, and biomarker led insights, we build representative virtual populations that help sponsors understand how a therapy may perform across different patient groups.
We also enable trial scenario simulation, allowing multiple study designs, inclusion criteria, and endpoints to be tested virtually before finalizing a protocol. This helps reduce uncertainty and supports more precise, efficient clinical planning.
At an advanced level, we are strengthening our capabilities across data science, biostatistics, and clinical pharmacology, while collaborating with validated computational platforms and technology partners. This allows us to move toward more refined patient level modeling and digital representations that can support personalized and indication specific insights.
Equally important, we ensure that all simulation outputs are aligned with clinical endpoints and regulatory expectations. We support validation against historical data, structured documentation, and reporting so that in silico evidence can contribute meaningfully to decision-making and future submissions.
What sets us apart is our ability to carry these insights forward. From protocol design and feasibility to clinical operations, data management, and regulatory strategy, we ensure continuity across every stage of development.
At RCS, we are not treating in silico trials as a future possibility. We are building and applying the capabilities required to make them work today, helping sponsors move forward with greater clarity, reduced risk, and stronger scientific confidence.
Challenges on the Road Ahead
While in silico trials are advancing rapidly, important challenges still need careful attention. Human biology remains highly complex, and the accuracy of any simulation depends on the quality of data and the assumptions used to build the model. Ensuring that these models are reliable, reproducible, and scientifically robust continues to be a key focus across the industry [6].
Regulatory acceptance is also evolving. Agencies are increasingly open to the use of computational evidence, but clear guidance, standardization, and consistent validation approaches are still developing. Demonstrating alignment between simulated outcomes and real clinical data remains essential for broader adoption [3].
In addition, considerations around data quality, integration of diverse datasets, and transparency of modeling approaches must be addressed. As these areas mature, in silico trials are expected to play a stronger role alongside traditional methods, supporting more efficient and informed drug development rather than replacing it entirely.
Final Thought
Drug development is entering a phase where better decisions are made earlier, with greater confidence and far less uncertainty. In silico trials are not just accelerating timelines, they are reshaping how therapies are understood, refined, and brought closer to patients.
At RCS, we see this as a defining shift in the future of clinical research. Our vision is to create a development ecosystem where simulation and clinical execution work seamlessly together, where every study begins with stronger evidence, clearer direction, and a deeper understanding of patient variability.
We are building toward a future where digital insights guide real-world outcomes, where risks are addressed before they reach the clinic, and where innovation translates into faster, safer, and more precise therapies.
The question is no longer whether in silico trials will influence drug development. It is how effectively they are integrated.
At RCS, we are committed to making that integration practical, reliable, and impactful for every program we support.
References
